Giúp e bài n với mn ơi e c.ơn ạ mong mn giúp e😢
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


1. What will you do?
I will organize free tutoring classes, donate books and school supplies, and raise funds for students in need.
2. Why do you choose to do them?
Because education is very important, and I want to help students have better opportunities to learn and succeed.
3. Do you think the students in need will receive benefits? Why?
Yes, they will. They can improve their knowledge, have enough materials to study, and feel more confident about their future.
4. How can you make more people know about your voluntary activities?
I can share information on social media, invite friends to join, and cooperate with schools or local organizations to spread the message.

Câu 2
\((1) MnO_2 + 4HCl \to MnCl_2 + Cl_2 + 2H_2O\\ (2) Cl_2 + H_2 \xrightarrow{as} 2HCl\\ (3) 3Cl_2 + 2Fe \xrightarrow{t^o} 2FeCl_3\\ (4) 2FeCl_3 + Fe \to 3FeCl_2\\ (5) 2NaOH + Cl_2 \to NaCl + NaClO + H_2O\)
\((1) 4Al + 3O_2 \xrightarrow{t^o} 2Al_2O_3\\ (2) 2Fe + 3Cl_2 \xrightarrow{t^o} 2FeCl_3\\ (3) C + O_2 \xrightarrow{t^o} CO_2\\ (4) 2KMnO_4 \xrightarrow{t^o} K_2MnO_4 + MnO_2 + O_2\\ (5) 4P + 5O_2 \xrightarrow{t^o} 2P_2O_5\\ (6) 2KClO_3 \xrightarrow{t^o} 2KCl + 3O_2\\ (7) Fe + H_2SO_4 \to FeSO_4 + H_2\\ (8) Cu + 2H_2SO_4 \to CuSO_4 + SO_2 + 2H_2O\\ (9) 2Fe + 6H_2SO_4 \to Fe_2(SO_4)_3 + 3SO_2 + 6H_2O\\ (10) 2Al + 6H_2SO_4 \to Al_2(SO_4)_3 + 3SO_2 + 6H_2O\)

a) \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right);n_{H_2SO_4}=0,1.2,5=0,25\left(mol\right)\)
PTHH: Zn + H2SO4 → ZnSO4 + H2
Mol: 0,2 0,2 0,2 0,2
Ta có: \(\dfrac{0,2}{1}< \dfrac{0,25}{1}\) ⇒ Zn hết, H2SO4 dư
b) \(V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c) \(m_{ZnSO_4}=0,2.161=32,2\left(g\right)\)
\(m_{H_2SO_4\left(dư\right)}=\left(0,25-0,2\right).98=4,9\left(g\right)\)
Bài 2 :
\(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
100ml = 0,1l
\(n_{H2SO4}=2,5.0,1=0,25\left(mol\right)\)
a) Pt : \(Zn+2H_2SO_4\rightarrow ZnSO_4+H_2|\)
1 1 1 1
0,2 0,25 0,2 0,2
b) Lập tỉ số so sánh : \(\dfrac{0,2}{1}< \dfrac{0,25}{2}\)
⇒ Zn phản ứng hết , H2SO4 dư
⇒ Tính toán dựa vào số mol của Zn
\(n_{H2}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
\(V_{H2\left(dktc\right)}=0,2.22,4=4,48\left(l\right)\)
c) \(n_{ZnCl2}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
⇒ \(m_{ZnCl2}=0,2.136=27,2\left(g\right)\)
\(n_{H2SO4\left(dư\right)}=0,25-0,2=0,05\left(mol\right)\)
⇒ \(m_{H2SO4\left(dư\right)}=0,05.98=4,9\left(g\right)\)
Chúc bạn học tốt

Bài 5:
a: \(A=\frac{-3\left(x+1\right)}{x^2-x-6}\)
\(=\frac{-3\left(x+1\right)}{x^2-3x+2x-6}\)
\(=\frac{-3\left(x+1\right)}{\left(x-3\right)\left(x+2\right)}\)
\(x^2-4=0\)
=>(x-2)(x+2)=0
=>x=2(nhận) hoặc x=-2(loại)
Khi x=2 thì \(A=\frac{-3\cdot\left(2+1\right)}{\left(2-3\right)\left(2+2\right)}=\frac{-3\cdot3}{\left(-1\right)\cdot4}=\frac94\)
b: \(B=\frac{2x}{x+3}-\frac{x}{3-x}-\frac{3x^2+9}{x^2-9}\)
\(=\frac{2x}{x+3}+\frac{x}{x-3}-\frac{3x^2+9}{\left(x-3\right)\left(x+3\right)}\)
\(=\frac{2x\left(x-3\right)+x\left(x+3\right)-3x^2-9}{\left(x-3\right)\left(x+3\right)}=\frac{2x^2-6x+x^2+3x-3x^2-9}{\left(x-3\right)\left(x+3\right)}\)
\(=\frac{-3x-9}{\left(x-3\right)\left(x+3\right)}=\frac{-3\left(x+3\right)}{\left(x-3\right)\left(x+3\right)}=\frac{-3}{x-3}\)
c: P=B:A
\(=-\frac{3}{x-3}:\frac{-3\left(x+1\right)}{\left(x-3\right)\left(x+2\right)}\)
\(=\frac{3}{x-3}\cdot\frac{\left(x-3\right)\left(x+2\right)}{3\left(x+1\right)}=\frac{x+2}{x+1}\)
Để P nguyên thì x+2⋮x+1
=>x+1+1⋮x+1
=>1⋮x+1
=>x+1∈{1;-1}
=>x∈{0;-2}
mà x là số tự nhiên
nên x=0

VIII
1 Have you ever been
2 Did you go
3 went
4 got up
5 have never got
6 got
7 Have you ever seen
D
I
1 B
2 A
3 D
4 A
5 C





 Giúp e bài 2 thôi ạ bài 1 e làm r ạ! Mong mn giúp e, e cần gấp ạ!
Giúp e bài 2 thôi ạ bài 1 e làm r ạ! Mong mn giúp e, e cần gấp ạ!
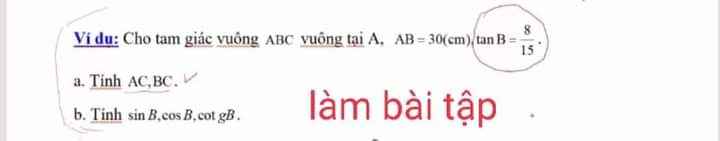



\(a,B=4\sqrt{x+1}-3\sqrt{x+1}+\sqrt{x+1}+2\sqrt{x+1}=4\sqrt{x+1}\\ b,B=8\Leftrightarrow4\sqrt{x+1}=8\\ \Leftrightarrow\sqrt{x+1}=2\\ \Leftrightarrow x+1=4\\ \Leftrightarrow x=3\left(tm\right)\)